Hi! Have you ever bumped into a glass door?
Many people have bumped into it, sometimes because the store's glass window wipe too clean, and the lack of obvious enough signs, but also make some customers hit. Because when you walk from a darker environment to a relatively bright area, it is not easy to find the middle of the blocked glass, it is most likely a very painful experience.

I also hit a glass door many years ago and broke the door of someone's factory floor. It was nighttime and they had lights in the workshop, but it was dark outside and there were no signs on the glass door, so I couldn't see the glass. Although the injury was not to the head but to the knee, but to pay people to match the glass still makes me heartbroken for a long time. However, I am much luckier than the next one.

See? It's a good thing that the smashed is tempered glass, so that the man did not suffer more serious injuries, otherwise the consequences are unthinkable.
Production of glass
Ordinary glass is widely used in our daily life, such as glass windows, mirrors, glasses and bottles and jars. The earliest glass was made by adding co-solvent to quartz sand and then fused at high temperature, and its main component was silica, so it was called quartz glass or silicate glass. It was gradually replaced by soda lime glass because it needed to be heated to a very high temperature, consumed a lot of energy and was costly.
The flat (float) glass used to make glass doors and windows and the glass used to make glass bottles contain slightly different proportions of minerals, in general they both contain roughly 73% to 74% silica, 13% to 14% sodium oxide, 9% to 10.5% calcium oxide, and the corresponding ratio of magnesium oxide, aluminum oxide, potassium oxide and other additives. When the quartz sand, soda, lime and dolomite and other raw materials are fully mixed and heated in the melting furnace at about 1500°C, these raw materials begin the chemical reaction of "vitrification" to produce a hot molten state of glass. After letting it flow into the tin bath to cool, it is made into flat glass, which can be poured into molds and blown into various bottles.

Flat glass is easy to process and can be mass-produced, so it is inexpensive and very widely used. Because it can both protect the wind and rain and allow light to pass through, it is very popular.
But flat glass also has an inherent disadvantage, that is, it is very brittle, a slight impact will break, and the glass fragments are extremely sharp, it is easy to hurt people.
People wanted to find a way to take advantage of the benefits of glass while making it less likely to break; even if it did break, it would not cause major damage to people. This is tempered glass.
Prince Rupert's Drop
When it comes to tempered glass, it is impossible not to mention the famous "Prince Rupert's Drop". In fact, it is a small glass ball, the shape of the head like a drop of water, while it has a long, thin tail, as a long drop of tears in general.

This "tear" looks ordinary, but in fact is not simple. Because its head has an incomparable strength, even the impact of a bullet can not make it break.
But this "Prince Rupert's Drops" has an Achilles' heel, that is, its long, slender tail, you just need to gently pinch its tail, the entire glass beads will be instantly reduced to pieces.

Why is that?
It starts with the birth of the "prince teardrop".
When we drop a molten glass drop into water, its surface will quickly become cold and solidified. However, the internal cooling of the glass bead is not so fast, so it is only the surface contraction hardened, the central part is still in the hot expansion of the state, so the volume of the entire glass bead does not reduce much. And when the glass in the middle of the droplet slowly becomes cold, its shell has been fixed, the glass in the center to shrink but is tightly pulled by the shell so that it can not shrink.

A strong stress is then created between the center of this glass bead and the shell. This stress pulls the network of silica molecules together so tightly that the otherwise fragile glass becomes so hard that it can resist pressures of up to 20 tons.

The toughened glass takes advantage of the mechanics of "Prince Rupert's Drop".
Production of tempered glass
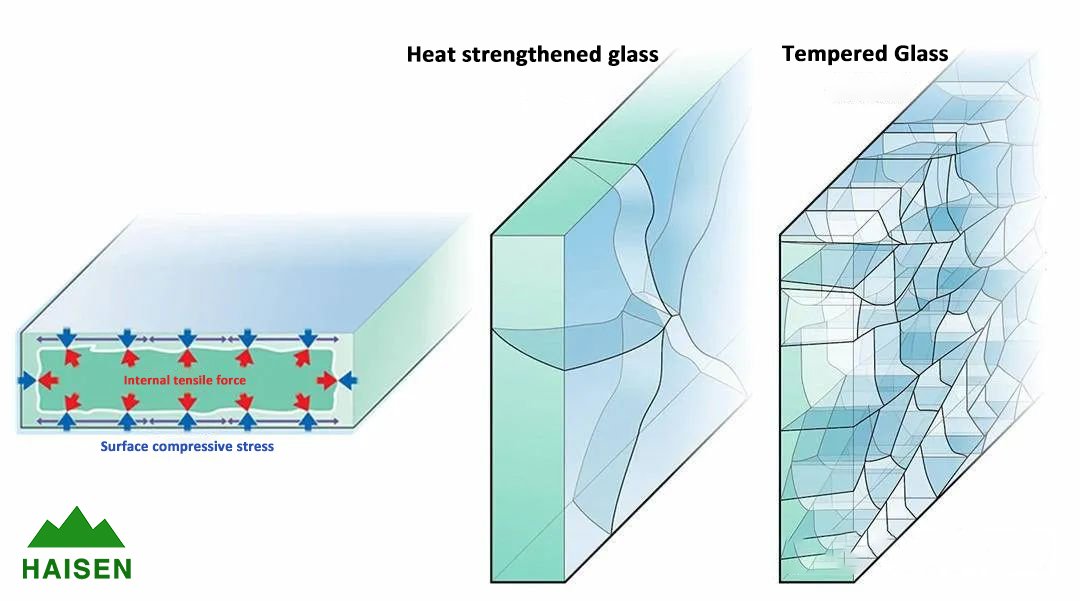
Tempered glass is made from ordinary glass through a heat treatment process known as "tempering". Tempering generates strong stresses that compress the surface of the glass and pull it internally, so that the stiffness and toughness of the glass are greatly enhanced. We can easily see the internal stress division of tempered glass by comparing the fracture of the glass after it is broken.

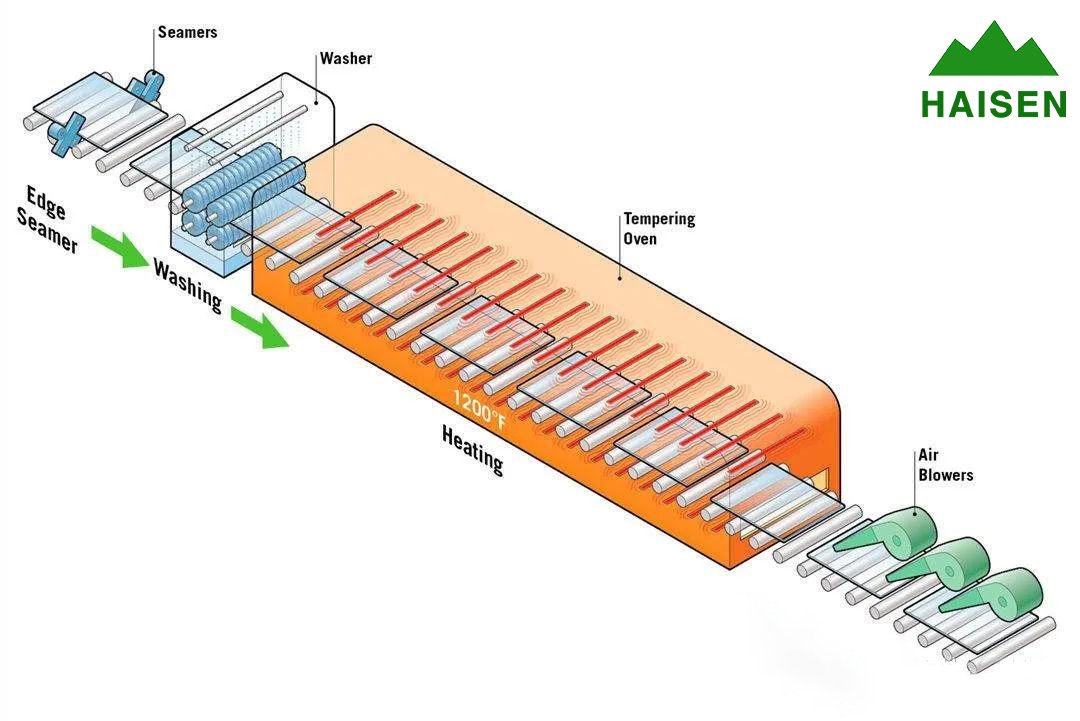
Before making tempered glass, workers need to cut ordinary glass into the shape and size required by the drawing, and polish the edges and corners, and drill all the holes that should be drilled, because all this can't be done after tempering.
After cleaning, the glass is heated in a long heating furnace to a temperature of over 650°C. At this time, the glass is softened and the stresses formed inside during the previous process are removed. When the conveyor belt sends the glass out of the furnace, the chillers arranged outside cool the surface of the glass evenly from all directions so that the outer layer hardens quickly, and then the inside of the glass cools down a little on the long conveyor belt.
Tempering causes a strong compressive stress on the surface of the glass, and this force is balanced by the tensile stress in the center of the glass. When the compressive stress on the surface of the glass is 3500 to 7500 psi (pounds per square inch), we call it heat strengthened glass; when the surface compressive stress is greater than 1000 psi, it can only break into small pieces when it breaks, and we call it tempered glass; only when the surface compressive stress is greater than 15000 psi (about 100 MPa), the edges of its pieces become less sharp, and this glass is called safety glass.
Why does tempered glass break into crumbs?
Due to the strong internal stress, the molecules of tempered glass are firmly bound together, which makes the glass stronger and more resilient than its previous fragility, and it will not be easily broken or crushed.
Likewise, it is due to the existence of a huge stress effect inside the glass, the whole glass, especially the corners are always in a tense state. Even if there is a small scratch or break on its surface, the stress will be concentrated on this point, breaking the original balance from the surface and causing the breakage of molecular bonds at the breakage. The energy of molecular bond breakage is transmitted inside the glass at the speed of sound, and the stresses collected inside the glass are released from the weakest point nearby, thus causing extensive molecular bond breakage. So in a flash, the whole glass shatters into crumbs. This is the reason that tempered glass cannot be worked on after it is made. Whether you try to drill a hole in it, hit it with a sharp object, or just take a glass cutter and scratch the surface, it can be immediately scrapped.

The degree of glass breakage is closely related to the amount of surface compressive stress previously formed during the tempering process; the greater the compressive stress, the more completely the glass breaks and the safer the pieces it forms. This is important because those shattered glass doors and windows or bathroom glass, as long as it exists sharp corners, can still cause skin scratches, and in severe cases can even cut through blood vessels.

In order to make the glass safer, people not only use heat-treated tempered glass, but also add a layer of polymer material film between the two layers of glass, so that even if it is shattered by impact, the pieces will not fall off and fly everywhere. This laminated safety glass is very common in cars, it has become the standard for windshields.

Conclusion
Glass is widely used in our life, it is easy to break and sharp fragments can cause injury, so the process of heat treatment has been used to improve the production of glass.
From the mechanical analysis of "Prince Rupert's Tears", we know that the rapid cooling of glass generates strong internal stresses, which make the glass stronger. Glass manufacturers then add a "tempering" process to produce tempered glass, which is stronger and safer.
When a defect occurs on the surface of tempered glass, the stress is concentrated in the defect and breaks the molecular bonds, and the energy of the break spreads rapidly in all directions.